Serology testing for IgM and IgG specific to SARS-COV-2
Serology testing for circulating IgM and/or IgG will be used to determine any prior exposure to SARS-COV-2 including those in the population that may be asymptomatic or patients who have recovered. Time course of IgG and IgM may provide insight to the immune response of patients to the infection. From a surveillance perspective, determining the status of circulating antibodies may provide ways to assess the COVID-19 pandemic.
The CDC has developed and validated a microneutralization test that can detect patient serum antibodies against SARS CoV-2 with 97% analytic sensitivity and 97% analytic specificity. And the FDA has given EUA to Cellex, Inc. for the first qualitative serology test for their qSARS-CoV-2 IgG/IgM Rapid Test that may be performed in moderate (and high) complexity CLIA compliant labs.
The challenge for serology testing resides in establishing panels of reference serum or plasma samples from patients to validate various assays to measure patient immune responses. The types of serology tests are listed eloquently in the table below from the Johns Hopkins website: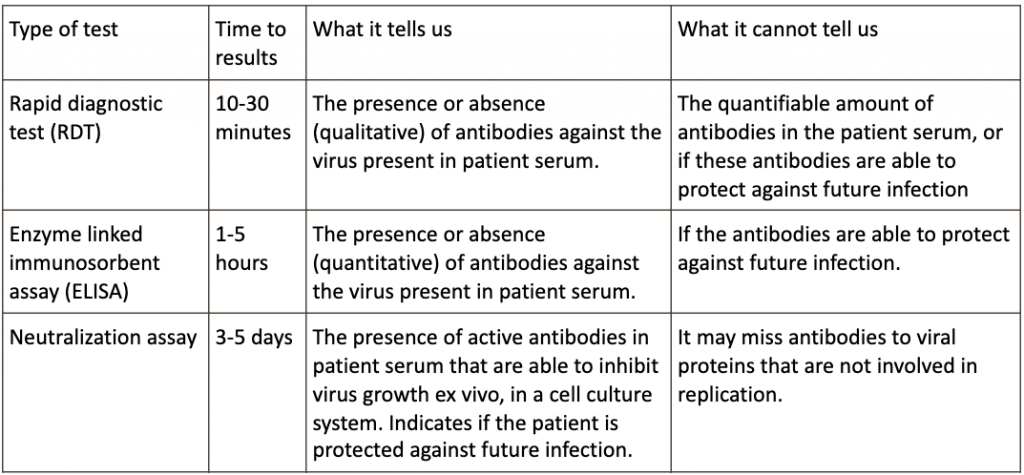
The links to each figure are here:
The FDA has issued a guidance to allow health care providers to treat eligible patients with convalescent plasma from eligible donors. Serology testing will play a pivotal role in this investigational mode of treatment.
Recordings of weekly CDC updates can be found at the link below along with written transcripts and sometimes the slides that are used as discussion points:
https://www.cdc.gov/safelabs/resources-tools/covid-19-weekly-clinical-calls/2020-march.html
Recordings of weekly FDA Town Halls can be found at the link below along with written transcripts and sometimes the slides that are used as discussion points. These Townhall meetings are mostly Questions and Answers with very little substantive presentation from the FDA: